Polarity influences extraction efficiency in branches, flowers, leaves,... | Download Scientific Diagram
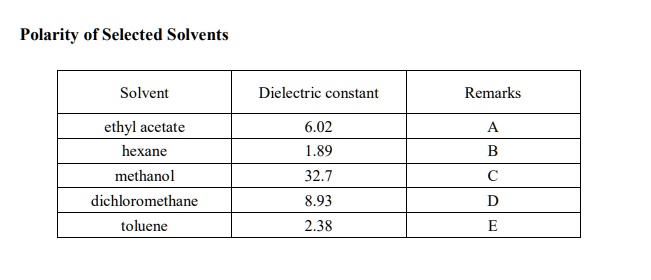
SOLVED: Polarity of Selected Solvents Solvent Dielectric constant Remarks ethyl acetate hexane 6.02 1.89 methanol 32.7 8.93 2.38 dichloromethane toluene

Question Video: Selecting the Statement Which Explains Why Hexane and Water Are Not Miscible | Nagwa
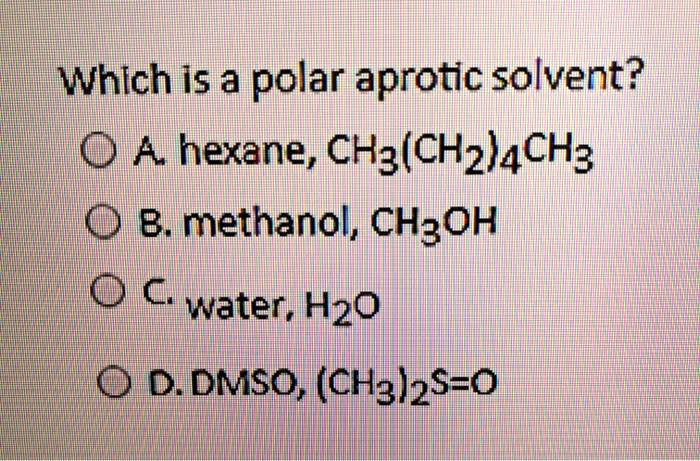
SOLVED: Which is a polar aprotic solvent? 0 A hexane, CHz(CHz)4CH3 B. methanol, CH3OH Cwater, Hzo 0 D. DMSO, (CH3l2S=0

Revision What is the symbol for slightly negative? Can non polar molecules have polar bonds? What are polar molecules? What are the three types of intermolecular. - ppt download