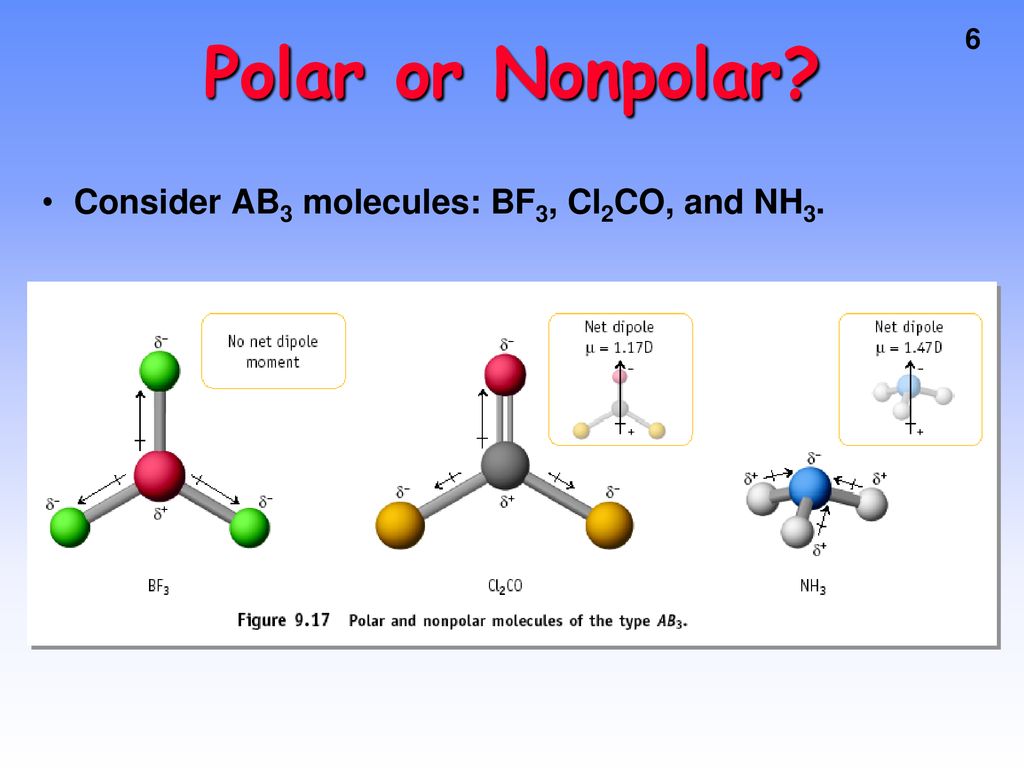
SOLVED: 41. The BF3 molecule is nonpolar; whereas the NF: molecule is polar: Which of the following statements accounts for the difference in polarity of the two molecules? In NFs, each F

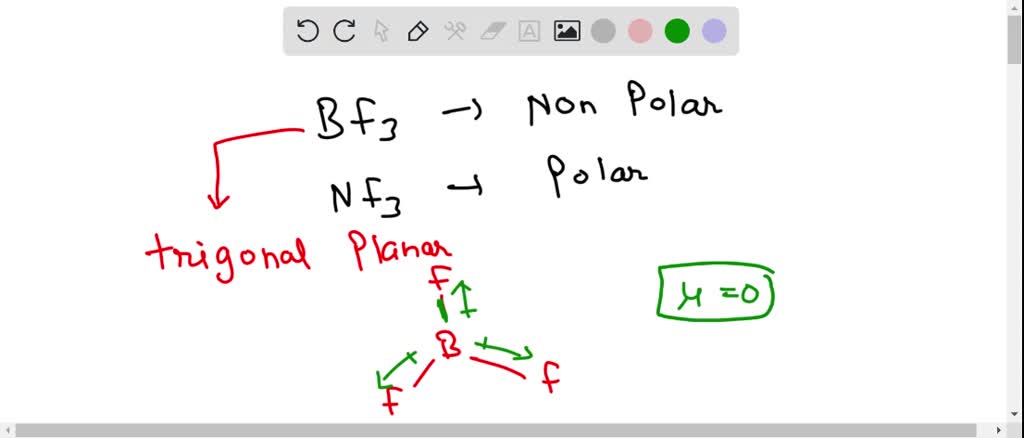
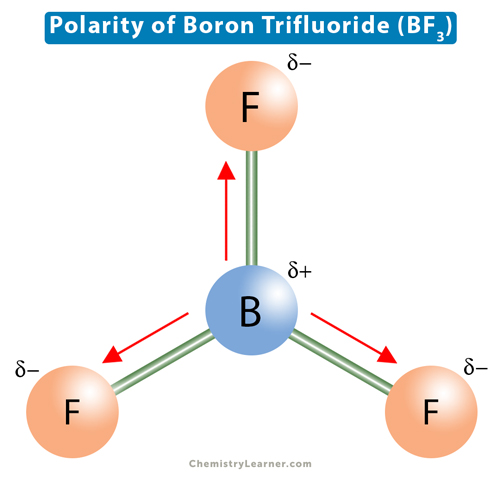
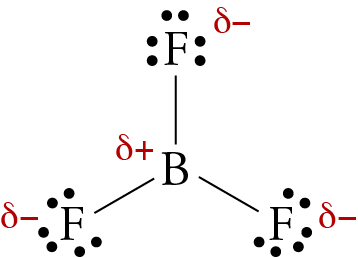
The molecule BF3 and NF3 , both are covalent compounds but BF3 is non - polar and NF3 is polar. The reason is that:

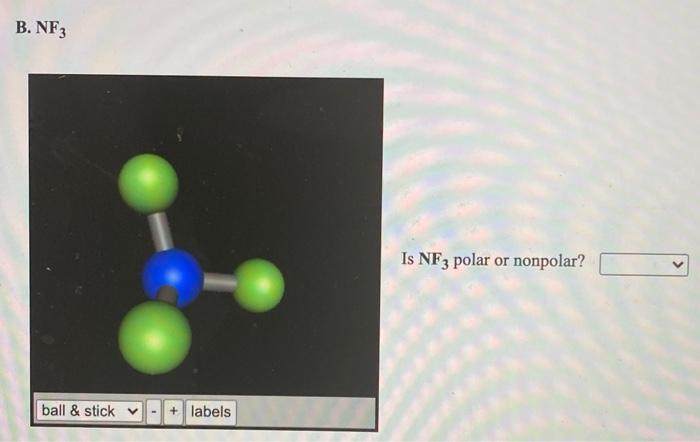
BF3 and NF3 both are covalent compounds but NF3 is polar whereas BF3 is non - polar. This is because :

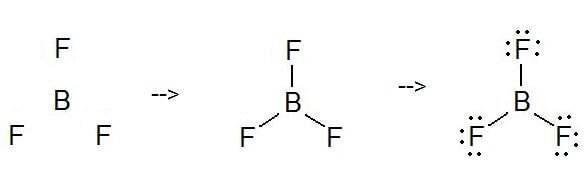
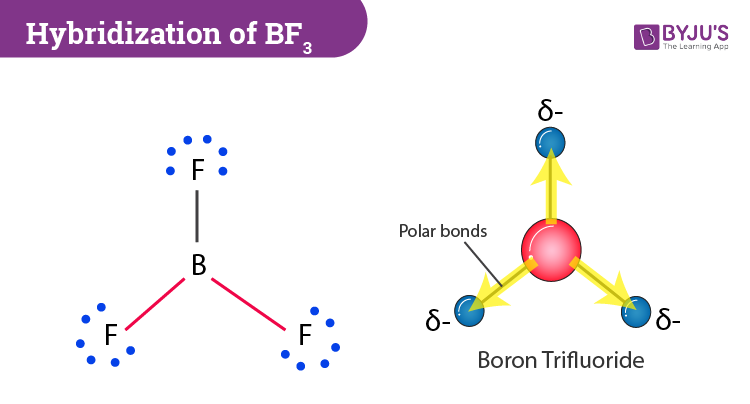
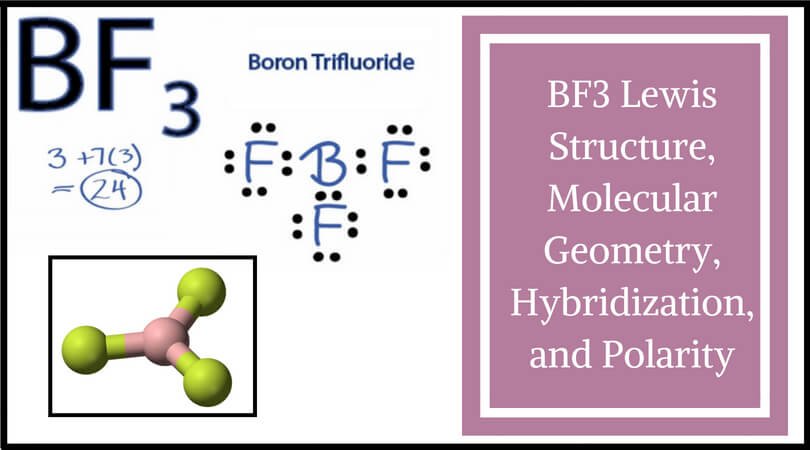
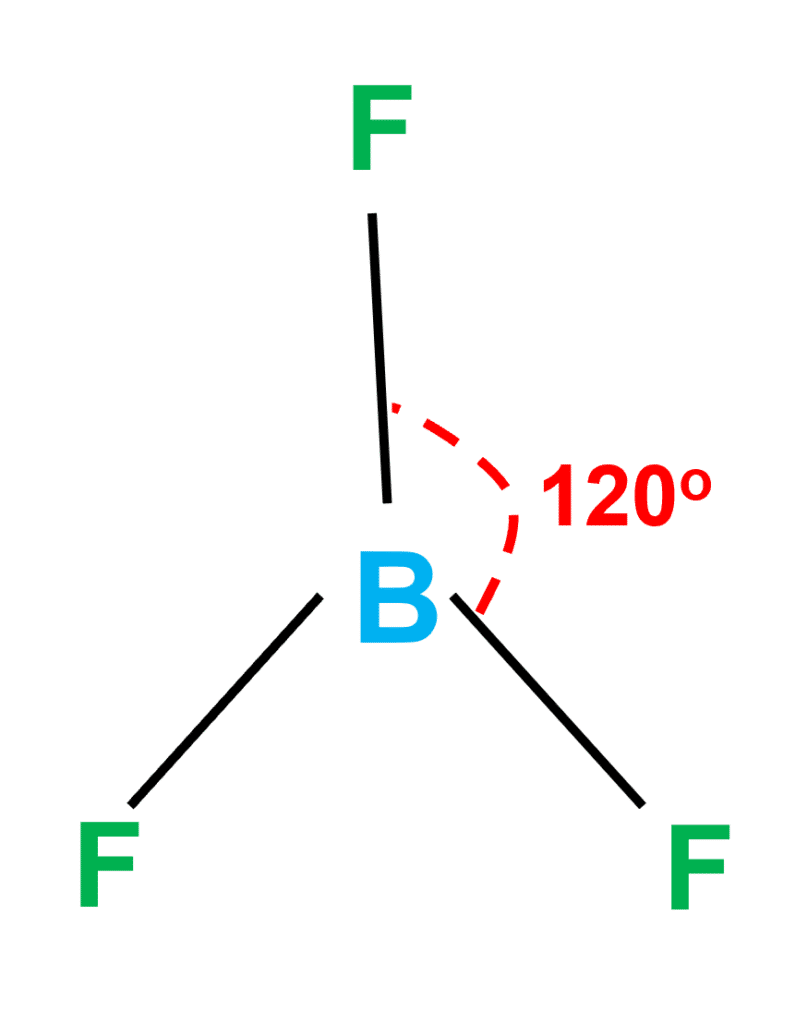
BF3 Molecular Geometry, Shape and Bond Angles (Boron Trifluoride) | BF3 Molecular Geometry, Shape and Bond Angles (Boron Trifluoride) Today in this video, we help you determine the molecular geometry of a
Ammonia (NH3) is a polar molecule while boron trifluoride (BF3), is a nonpolar molecule. What is the difference in the polarity of these compounds? - Quora